Structure of the Commission
The Commission is organised into 15 working groups which take on concrete work packages relating to the risk assessment of substances, operating in close coordination with each other.
The main focus is on the work carried out by the “MAK Values” Working Group and the “Assessment Values in Biological Material” Working Group, where the proposed threshold limit values are developed for the MAK and BAT values. The “Air Analyses” Working Group and the “Analyses in Biological Materials” Working Group develop the relevant measurement methods that are indispensable for compliance with the values. As a supplement to the threshold limit values, specific classifications of the substances with regard to their carcinogenic, allergic, skin-absorbing and germ-line-related effects are identified in the relevant working groups and the results are passed on to the central working groups. In the case of particularly complex and relevant groups of substances such as dusts, there are additional working groups that pursue work on generic effect mechanisms in greater depth. Current topics such as the role of epidemiological studies for evaluation in the low dose range are addressed by dedicated, temporary working groups.
Once a year, all working groups come together for a plenary session to conclusively discuss the compiled results and formally approve the list of MAK and BAT values.
Reports from the plenary sessions of the MAK Commission:
(interner Link) I (interner Link) I (interner Link) I (interner Link) I (interner Link)
The work of the Commission would be unthinkable without the input and organisational support provided by the Commission’s Scientific Secretariat. Its tasks include extensive research and the preparation of texts as a basis for decision-making.
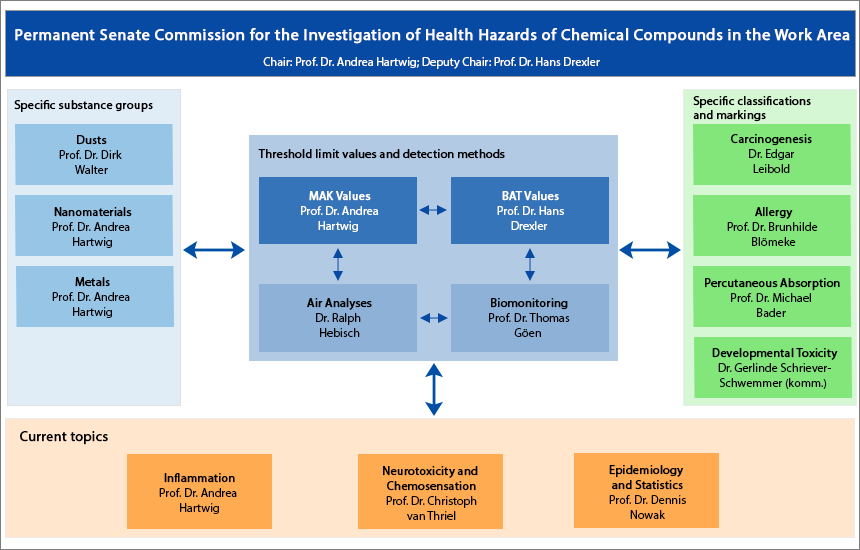
If you would like to know more about the composition and tasks of the working groups, click on the respective working group in the graphic above. (interner Link)
The Commission’s Scientific Secretariat
The Commission’s Scientific Secretariat organises and coordinates the work of the Commission and is responsible for content review, editorial processing and publication of the List of MAK and BAT Values and the scientific substantiations. It participates in conceptual work such as the development of new evaluation criteria for carcinogenic, germ cell mutagenic or allergy-causing substances and liaises with other expert committees, both national and international. As such, its support ensures the working capacity of the Permanent Senate Commission for the Investigation of Health Hazards of Chemical Compounds in the Work Area.
Management:
- Dr. Gunnar Jahnke
- Dr. Gerlinde Schriever-Schwemmer
Karlsruhe Institute of Technology (KIT) Institute for Applied Biosciences,
Department of Food Chemistry and Toxicology
MAK Commission
Adenauerring 20a
Building 50.41, Room 105
76131 Karlsruhe
Tel. +49 721 608-47400
E-mail: (externer Link) - (Download)
- (Download)