How the Commission operates
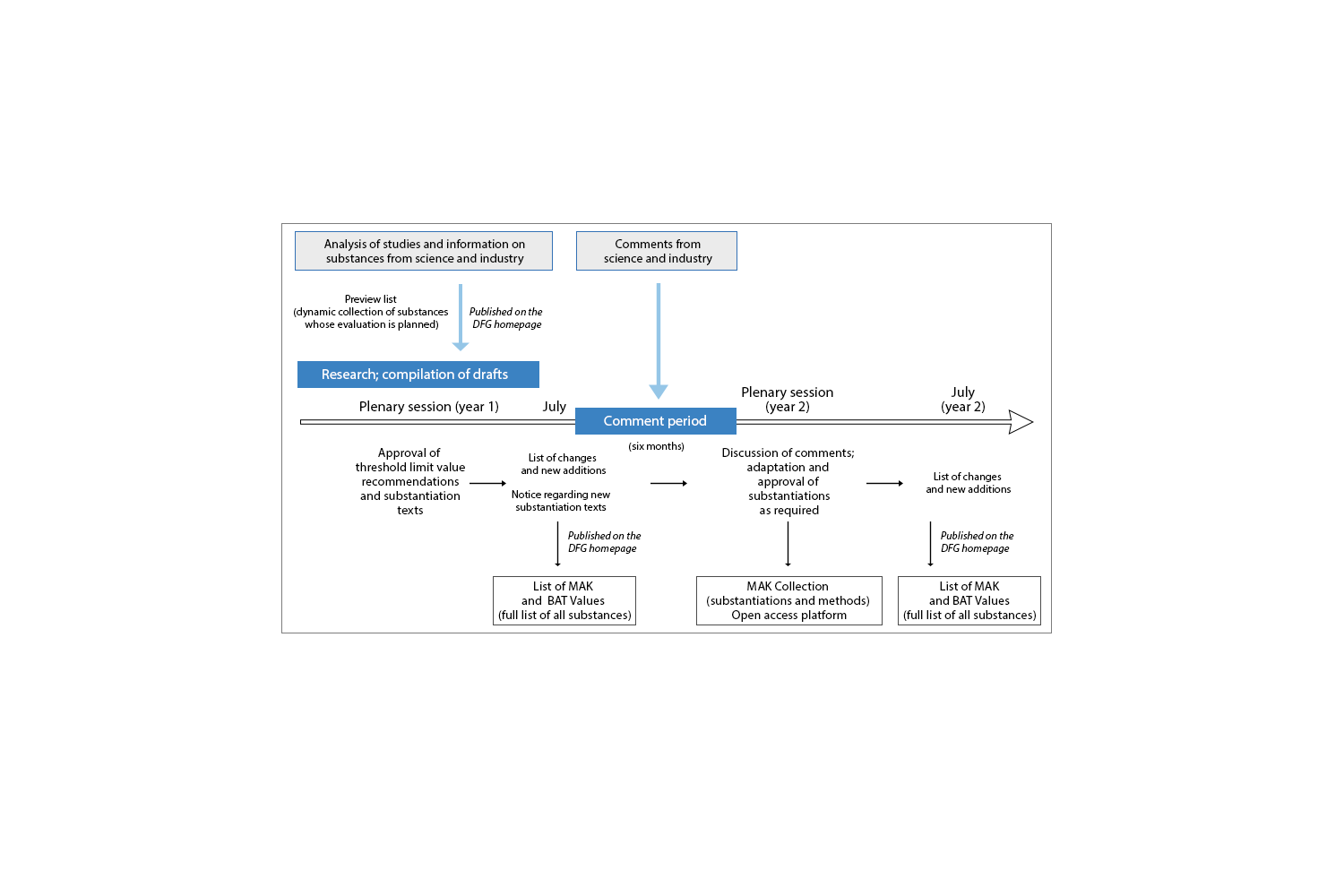
In order to enable a proposal to be developed for a threshold limit value, the process begins with an extensive literature and knowledge review supported by the Commission's Scientific Secretariat, with the aim of compiling all findings relevant to a given substance. On this basis, and following a qualitative review of all studies, a proposed threshold limit value is developed wherever possible. In all cases, the current state of knowledge on the toxicological properties of a substance is systematically summarised and published in the form of a scientific substantiation. Where significant new findings emerge, these substantiations are revised and the threshold limit value is adjusted if necessary. In addition to the substantiations, information is also published on the measurement methods by which the threshold limit values can be determined.
The results are documented in detail, thereby ensuring that they are transparent and verifiable. The process also ensures that the public is adequately involved.
One way of supporting the work of the Commission is to submit information and observations from practical experience relating to a substance, or studies that have not yet been taken into account. To facilitate this, a preview list is published (see below) that identifies the substances for which reviews have been initiated.
At the Commission’s plenary session, which takes place in spring, threshold limit value recommendations that are ready for approval are formally approved along with the relevant substantiations. A list of amendments and new entries (see below for the current edition) is published every July as part of the annual List of MAK and BAT Values. This is followed by a six-month comment period, during which critical comments, questions or ambiguities may be submitted to the Commission. The comments are discussed and responded to, and classifications or substantiations are revised and amended where appropriate. Not until this process is complete are the results formally approved and posted on the open-access platform – the so-called MAK Collection. If new findings or studies emerge, however, the discussion of a substance can be re-initiated again at any time. The classifications are derived from a detailed description of the categorisation and nomenclature, which is provided in the List of MAK and BAT Values or in the MAK Collection.
In addition to the classification of substances, the Commission also describes measuring methods and publishes them in the MAK Collection, thereby ensuring that it is possible to comply with threshold limit values. The measurement methods are tested and confirmed by at least one independent laboratory prior to publication.
The Commission is essentially free to (Download). However, suggestions from practical experience or proposals for substance or method analysis can be taken up at any time.